
CLINTEL: Data-driven clinical trials monitoring
CLINTEL (formerly Advanced Bio-Logic Solutions) identified a growing need in drug development: enabling clinical development organizations to comply with FDA draft guidance on risk-based monitoring principles. CLINTEL saw the potential to develop easy-to-use tools that could also improve quality and significantly reduce costs.
“A typical phase III trial costs between $80-100 million,” says Jeff Epstein, CLINTEL’s CEO. “The industry estimates that up to 30 percent of clinical trial costs are related to site monitoring. Any reduction to site monitoring costs has the potential for significant impact, especially if you can increase quality along the way.”
Focusing on What Matters
In August 2013, the FDA finalized guidance on the topic of a “risk-based approach” to monitoring clinical trials. In it, the FDA advocates alternatives to regular, frequent onsite visits and verification of all data, which involve costly travel and provide little added benefit. Instead, they recommend approaches that identify risks to patient safety by utilizing critical data elements gathered in clinical data systems as a method of determining which sites warrant additional attention from clinical research associates (CRAs). Ultimately, this allows sponsors to better focus their monitoring efforts, sending CRAs to the sites whose risk level warrants more attention.
"The FDA advises that companies engaged in trials, specifically, the biopharmaceutical and medical device industries and their development organizations, should focus on what matters: risks to patients and risks to study data," Epstein says. "But identifying those risks can be difficult."
Since clinical trials are designed to collect important data about a potential drug’s safety and efficacy, there’s no shortage of data that can also be used to assess risk. The problem lies in getting that information to the right people at the right time, and in their language.
Silos and Gaps
The CLINTEL team, with its decade of experience offering electronic data collection system services for companies engaged in the clinical trials process, knew that an effort to enable this sort of analysis to clinically-focused users would face some distinct challenges.
Jon Denne, Director of Business Development, explains, “From executives to researchers, people need access to information in an actionable format. Systems capture the information, but most don’t really have an analytics toolset that shows the entire picture.”
“Excel might be the most widely used report engine in Pharma,” Epstein adds. “The biggest risk, which I’ve seen firsthand, is the risk of inaccuracy. And when people are basing decisions on incorrect data or inconsistent data—that problem tends to proliferate.”
Despite these issues, spreadsheets have become the default report technology for many business users because they have simply had no other option.
“There are tools out there that are good at providing bio-statisticians flexible insight into clinical data, but study management teams are often left to access data from several different systems and make sense of it on their own. And then they stitch it all together in spreadsheets,” Epstein says.
Using spreadsheets as the go-to tool for the non-programmer carries inherent hazards. In addition to accuracy and consistency issues, the presentation of data also can be a challenge. Spreadsheets can be impossible to digest at a glance, and slides or dashboards with 15 or 20 pages risk overwhelming the audience.
“When you present too much information, you see almost nothing,” notes Adnan Shawkat, CLINTEL’s Director of Information Systems. “There’s Excel and then there’s SAS,” explains Shawkat, referring to the ubiquitous statistical analysis tool. “Unless you're a statistical programmer, there’s nothing in the middle.
Evaluating the Market
In addition to Tableau, CLINTEL considered several other business intelligence tools. This included Microsoft Reporting Services, which CLINTEL could have used for no additional cost as part of its Microsoft SQL Server licensing.
“We evaluated many solutions in the market, but didn't find any as comprehensive as Tableau,” says Shawkat.
The trend among them was bespoke solutions that required costly consulting services. While CLINTEL is willing and able to adapt its services for those companies that want to leverage their existing investments in other reporting technologies, they decided against those software packages for its go-to offering.
"Most solutions require extensive configuration. There isn't much pre-built, out of the box,” says Denne. “Our goal is to offer an analytics portfolio that can stand on its own, but is highly configurable based on customer requirements. Ultimately this will shorten the software development cycle and reduce associated costs.
In order for a solution to be viable it has to integrate with existing infrastructure effectively. As a core requirement, an analytics portfolio needs to be consistent, actionable, and readily available to all members of a clinical project management team.
“We evaluated the market, and Tableau has the strongest capabilities in several areas. We liked the aesthetics of the visualizations—it’s clean, it’s easy to use, and is ideal for a non-statistical end user who needs to access information quickly. We liked Tableau's functionality and ability to connect to disparate data sources. We especially liked the Tableau data engine.” says Denne.
From Idea to Reality
CLINTEL has developed a data analysis solution and services set that is currently rolled out to several customers conducting phase II and phase III studies.
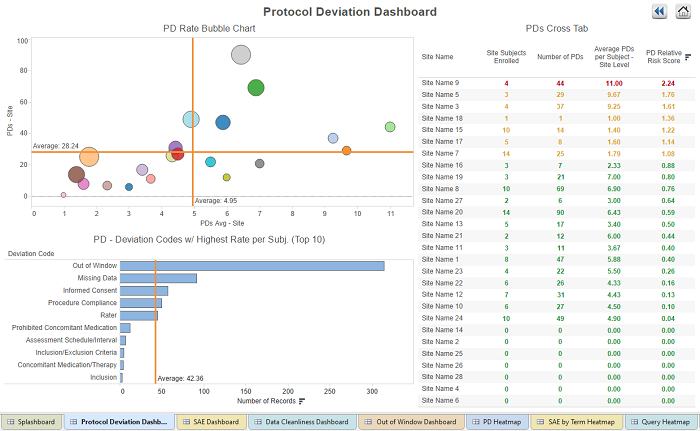
Figure 1: Dashboard showing site performance within the protocol deviation category
CLINTEL’s Tableau implementations connect to multiple data systems, including clinical trial management systems (CTMS) and electronic data capture (EDC) systems.
“The great thing is that this solution is source agnostic. Your study could be set up using CTMS or EDC from Oracle, Medidata, BioClinica or any one of the companies who provide these source systems,” says Shawkat.
The ability to connect to cloud data sources also plays a big part in the solution. CLINTEL is currently working with customers who prefer the cloud-based model as it limits their maintenance and equipment burden.
"Providing a cloud option is essential to our offering. It allows access to key reporting technologies without requiring customers to make substantial investments into expensive server tools and technologies,” says Epstein.
CLINTEL analyzes customer data, creating Tableau visualizations that they can embed into customers’ SharePoint sites. User access is managed through Active Directory or Trusted Tickets, depending upon the customer’s preference. The solution is secure and 21 CFR Part 11 compliant.
"The Possibility Exists Where It Did Not Previously"
“We looked at everything, and for what we’re trying to help our customers accomplish, Tableau is the easiest, most accessible and aesthetically-pleasing solution. And it’s also going to be the most cost-effective,” concludes Epstein.
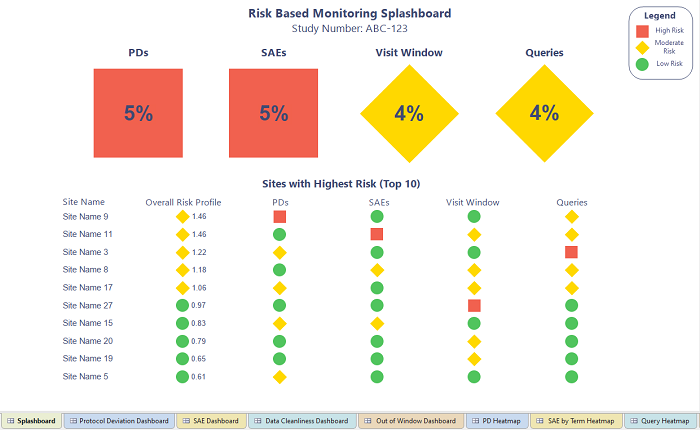
Figure 2: Splashboard representing the total health of the study
In particular, CLINTEL is pleased to note that with Tableau, it is able to:
• Negate data silo issues. The siloed data problem has been overcome with Tableau’s easy connection to disparate data sources. “Any repository where they’re collecting clinical information is what we’re going to connect to with Tableau,” says Denne.
• Close the reporting gap. With Tableau, CLINTEL is providing self-service analysis that hits the “sweet spot” between Excel and SAS. “Tableau makes the information that our customers already have accessible in an accurate way to people who otherwise wouldn’t have access without great expense and specialized skills,” says Epstein. Leveraging the advanced reporting capabilities of Tableau, CLINTEL is able to create dynamic visualizations that deliver critical data points to the correct audience.
• Increase speed-to-solution. Compared with the other data visualization tools out on the market, CLINTEL is able to produce solutions for its customers much more quickly using Tableau. “Tableau can get up and running a whole lot faster,” Denne says. Tableau provides a solid engine for data visualization and can manage large and complex data sets. This has enabled CLINTEL to offer a robust visualization tool that will allow clinical teams to make more accurate decisions much earlier in the drug development process.
• Create opportunities for savings. CLINTEL can see that its customers will be able to perform interim analysis on study sites much earlier, reducing risk and eliminating costs at under-performing sites. “A decision to cut a site can save you $100,000. A decision to complete an interim analysis to revise your protocol could save you a whole lot more because it’s affecting your entire program,” says Epstein. “Now the possibility exists where it did not previously.”
• Improve accuracy and enable insights. The easy-to-understand visualizations are helping customers avoid the inaccuracies and missed insights that were part of the Excel-based reporting world. “We can present complex, disparate data in a way that’s easier for people to digest,” says Shawkat.